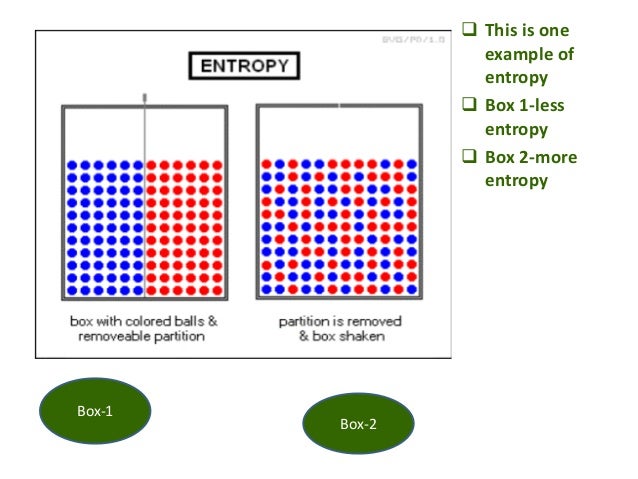
However, as calculated in the example, the entropy of the system of ice and water has increased more than the entropy of the surrounding room decreased. The entropy of the room has decreased and some of its energy has been dispersed to the ice and water. Over time the temperature of the glass and its contents becomes equal to that of the room. As shown in the ice melting example below discussing the illustration involving a warm room (surroundings) and cold glass of ice and water (system), the difference in temperature begins to be equalized as portions of the heat energy from the warm surroundings become spread out to the cooler system of ice and water. In a thermodynamic system, a "universe" consisting of "surroundings" and "systems" and made up of quantities of matter, its pressure differences, density differences, and temperature differences all tend to equalize over time. 4.1 Entropy increase as energy dispersal."Ice melting" - a classic example of entropy increasing The statistical definition of entropy is generally thought to be the more fundamental definition, from which all other important properties of entropy follow.Īlthough the concept of entropy was originally a thermodynamic construct, it has been adapted in other fields of study, including information theory, psychodynamics, and thermoeconomics. In terms of statistical mechanics, the entropy describes the number of the possible microscopic configurations of the system. In a simplified approach for teaching purposes, entropy is described as measuring the spontaneous dispersal of energy at a specific temperature, although this dispersal is not necessarily spatial.
Entropy examples free#
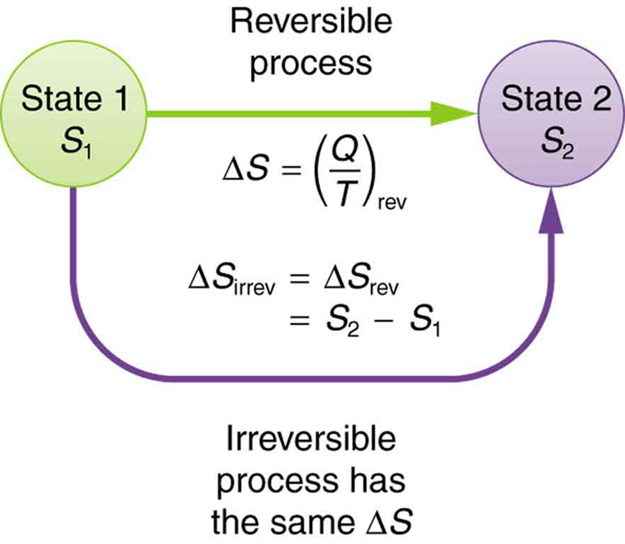
Entropy is one of the factors that determines the free energy in the system and appears in the second law of thermodynamics. In thermodynamics, entropy, symbolized by S, is a state function of a thermodynamic system defined by the differential quantity, where dQ is the amount of heat absorbed in a reversible process in which the system goes from one state to another, and T is the absolute temperature.

